Introduction
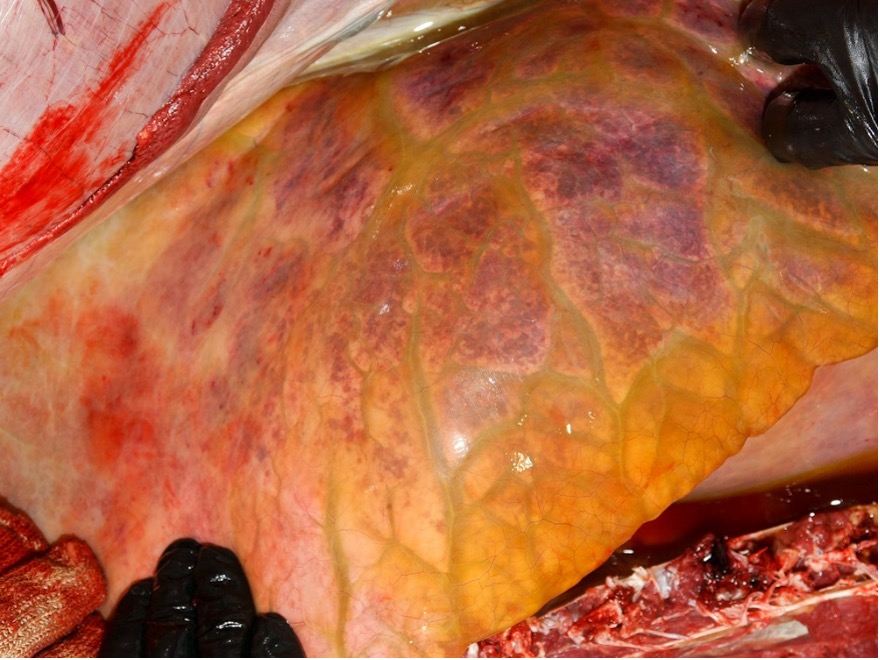
Endocrine dysfunction associated with obesity is a daily challenge in equine ambulatory practice and obesity-related laminitis poses a significant threat to equine welfare in Australia (Figure 1). A recent pony club survey in Victoria found that 15% of horses and ponies had suffered from at least one episode of laminitis and more than half of the affected animals had experienced multiple episodes [1].
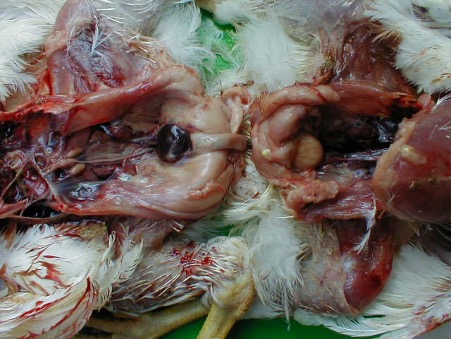
Equine metabolic syndrome (EMS) describes the syndrome of obesity, hyperinsulinemia and laminitis and accounts for most cases of laminitis in equine practice. Studies demonstrating that laminitis can be induced by protracted increases in insulin concentration [2,3] were pivotal in understanding that hyperinsulinemia is central to the development of laminitis (Figure 2). These have been followed by work in field cases demonstrating that the pathological changes that occur in both equine metabolic syndrome and pituitary pars intermedia dysfunction (PPID) are consistent with those that occur in laminitis that is induced experimentally with hyperinsulinemia [4,5].

Pituitary Pars Intermedia Dysfunction (PPID) has historically been associated with laminitis; however, its importance as a cause of laminitis remains to be fully elucidated as does the mechanism that might link PPID and hyperinsulinemia. Currently, it is debated whether EMS may predispose a horse to PPID and whether the two conditions are related [6,7]. Whilst the link between PPID and laminitis may have been overstated in the past, PPID remains a very common endocrinopathy of ageing horses and can result in a range of clinical signs that compromise welfare. Relative to EMS and PPID, other equine endocrinopathies are rare. Benign adenomas of the thyroid gland are seen occasionally but other disorders of the thyroid, parathyroid or adrenal glands are very rare.
Updates on the diagnosis of Equine Metabolic Syndrome
Equine metabolic syndrome is a collection of risk factors for endocrine laminitis with insulin dysregulation being the central feature. Specifically, it is persistent hyperinsulinemia that is thought to result in laminitis and investigation of EMS focuses on assessment of insulin concentrations.
Laboratory investigation may not be necessary – it is reasonable to assume that a young, fat native pony with laminitis is suffering from EMS and a laboratory diagnosis is unlikely to change the treatment recommendations that are made at the outset.
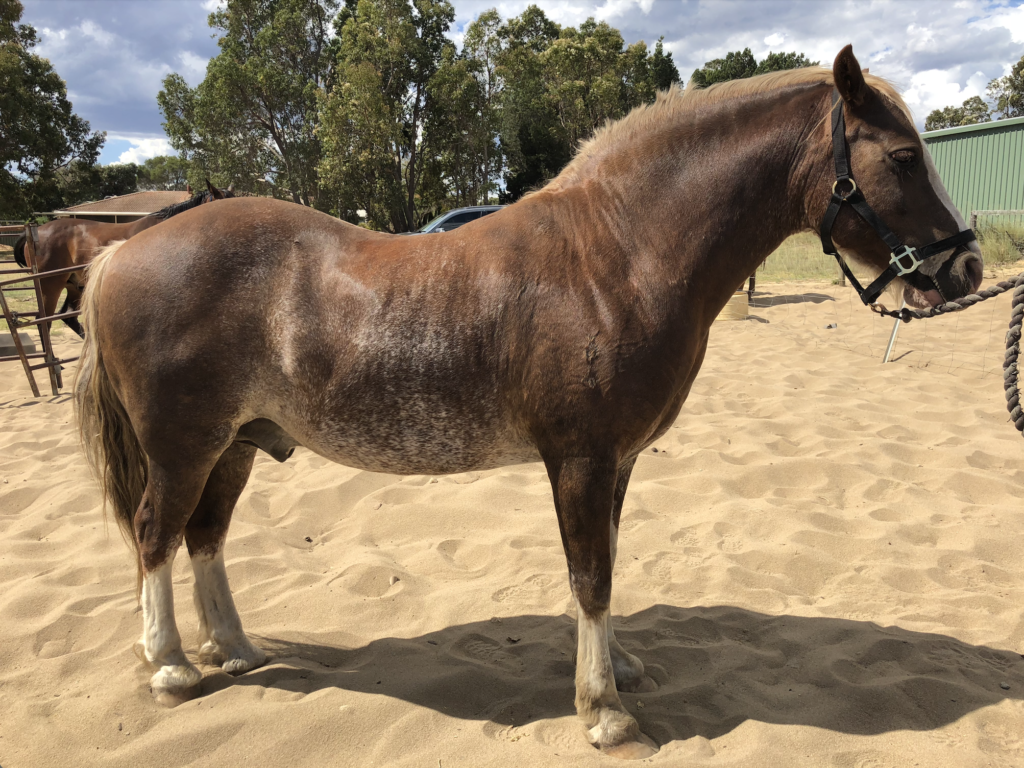
However, not all horses with EMS exhibit the typical obese phenotype (Figure 3) and assessment of hyperinsulinemia as well as measurement of adiponectin (not commercially available in Australia currently) and triglyceride concentrations may provide clinically useful information. Insulin concentrations can be assessed following a period of fasting, following feeding, following oral ingestion of a known quantity of sugar or following intravenous infusion of glucose and/or insulin.

These different tests have advantages and disadvantages. Intravenous tests are less convenient and are therefore unlikely to be performed in ambulatory practice but can be helpful in demonstrating tissue insulin resistance that may not be demonstrable using other tests. Measurement of serum insulin concentration without any form of sugar challenge is the most straightforward test in practice but false negative results are common. Due to its low sensitivity for identifying insulin dysregulation, fasting before measuring insulin concentration is no longer recommended [8]. Unexpected negative results when measuring basal insulin concentration should be followed-up with some form of sugar challenge test.
Measurements taken following feeding with forage are more variable but are more sensitive than basal insulin concentrations and are potentially more helpful as they give an indication of the insulin responses (and hence laminitis risk) that are occurring under normal feeding and management conditions. Stress from acute laminitis, transportation and management changes may also affect insulin sensitivity and this should be taken into account when interpreting test results [6,8]. Recent review articles provide further discussion of different options for laboratory investigation of EMS [9,10].
Updates on the Management of Equine Metabolic Syndrome
Surveillance data of horses and ponies in south-east Australia demonstrated the prevalence of researcher-assessed obesity to be 24%, with ponies, particularly Shetlands showing a higher prevalence of obesity [11]. Owners in this study also perceived animals to be in significantly lower body condition compared to researcher assessments.
The increasing prevalence of obesity in horses has paralleled rises in obesity in other domestic species, including our own, with the Australian Institute of Health recognising that 67% of adults in this country are either overweight or obese. Dietary management and increased exercise (where appropriate) should be the focus of efforts to improve tissue sensitivity to insulin and hence reduce the risk of laminitis, with pharmaceuticals being a secondary, but potentially important, consideration.
It should, in theory, be straightforward to manage the weight of our domestic animals by manipulating their dietary intake; however, this overlooks the complex behavioural and psychological factors that influence the relationships we have with our pets. Therefore, strategies are often required that can accommodate some of the barriers that cannot be broken down and would otherwise prevent weight loss and reduction in laminitis risk. The use of pharmaceuticals as a treatment for obesity and insulin dysregulation has traditionally been a last resort but as evidence increases for the potential welfare benefits of accelerated weight loss and reduced laminitis risk, so the use of these drugs has increased. In a recent study of UK native ponies, the risk of laminitis occurring within 3 years was 9.9% [12]; the risk in those that had a fed insulin of > 21.8 u/l was a staggeringly high 21.5%. With the risk of laminitis being so high in certain populations it seems entirely appropriate that a more pro-active approach to management is taken to prevent unnecessary suffering. In severe cases or if insulin cannot be reduced within a couple of months through management alone then the use of pharmaceuticals to reduce insulin concentrations (and potentially adiposity) should be considered. There are also anecdotal reports of the benefits of insulin sensitising drugs in horses with acute laminitis and hyperinsulinemia.
Ertugliflozin/Canagliflozin/Velagliflozin
Sodium glucose co-transport-2 (SGLT2) inhibitors have become a popular treatment for human type-2 diabetes mellitus and show considerable promise as a means of limiting hyperinsulinemia and reducing laminitis risk in horses. Through inhibiting re-uptake of glucose in the kidney and thereby promoting its excretion, insulin concentrations are lowered.
The risk of laminitis has been shown to reduce following the administration of velagliflozin [13,14]. Canagliflozin has also been shown to be effective in reducing insulin concentrations in horses with insulin dysregulation [15]. Ertugliflozin (Steglatro : Merck Sharpe & Dohme) is the newest SGLT2 inhibitor developed in human medicine and is available in Australia in 5mg and 15mg tablets. Like velagliflozin and canagliflozin, ertuglflozin has been shown to reduce insulin concentrations rapidly in horses with EMS (Tania Sundra, unpublished data) and its use has also been associated with rapid reductions in insulin concentrations and rapid clinical improvement in ponies with both acute and chronic laminitis.
Ertugliflozin has been used at 0.05 mg/kg PO SID. SGLT2 inhibitors have the potential to cause hepatic lipidosis in horses due to mobilisation of triglycerides from lowered blood glucose concentrations and urinary tract infections due to glucosuria, although none of these adverse effects have been noted in studies performed to date. Larger clinical field trials are required but all of the work performed thus far suggests that the SGLT-2 inhibitors could be very useful in both preventing and treating laminitis associated with hyperinsulinemia.
Levothyroxine
With the availability of a suitable palatable paste for horses in Australia (Bova Aus), levothyroxine has become increasingly popular for obese horses that are refractory to weight loss (Figure 4).
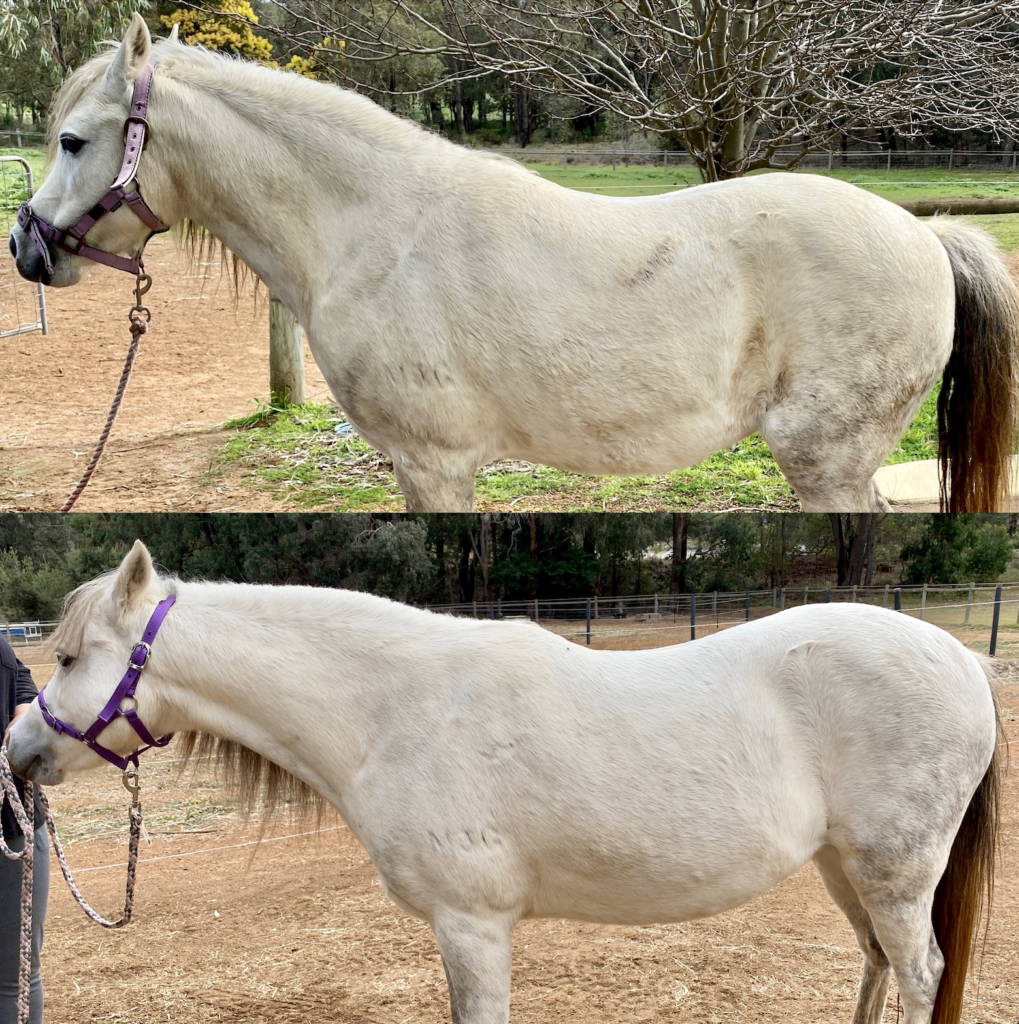
The administration of levothyroxine to obese horses with a history of laminitis results in both weight loss and improvements in insulin sensitivity compared to controls [16]. In healthy horses, levothyroxine administration is associated with a reduction in insulin concentrations, a doubling of insulin sensitivity, an increase in insulin disposal rate, a decreased insulin response to glucose and an increase in insulin receptors on adipocytes; concentrations of triglycerides, cholesterol and low-density lipoproteins also decrease [17–19]. The effects on insulin dynamics coupled with the promotion of weight loss make levothyroxine a compelling treatment option for obese horses that are suffering from, or are deemed to be at risk of, laminitis. In the USA where levothyroxine has been widely available at a more reasonable cost the drug has been used safely and effectively in the management of equine metabolic syndrome for decades. There is an increasing tendency to use levothyroxine in the treatment of acute laminitis in addition to using it as a treatment for obesity. Whilst evidence is lacking, this is logical considering the drug’s effects on insulin dynamics [20]. A number of investigations have now demonstrated that levothyroxine is safe and well tolerated even when used for very long periods or at extremely high doses [19,21,22]. Levothyroxine is typically used at 0.1 mg/kg PO SID, however anecdotally lower doses may be effective. Historically 3-6 months of treatment has been recommended but this is an arbitrary figure and the drug should be used until target weight is reached at which time the dose should be reduced gradually over a few weeks. Where finances do not permit 3-6 months of treatment, shorter courses can be helpful in promoting weight loss and in encouraging owners that it is possible to effectively reduce the weight of their horses. The authors tend to treat for one month at a time, re-assessing the clinical response before prescribing a further month of treatment. Levothyroxine will also stimulate appetite and horses prescribed this medication should be placed on a controlled diet in order to achieve weight loss. Monitoring of body condition is important as responses are variable, some horses will lose weight rapidly; others may not lose weight if there is no compliance with management recommendations. When target weight is reached it is helpful to assess insulin concentrations to determine whether there is persistent insulin dysregulation that may need to be managed indefinitely.
Metformin
Metformin became a popular treatment for horses with recurrent “pasture-associated” laminitis in the absence of other alternatives in Australia [23]. Its popularity has waned with the availability of more effective alternatives and with a growing perception that it does little to reduce the risk of laminitis. Evidence generated to date suggests that metformin acts at an intestinal level inhibiting the absorption of monosaccharides and in doing so reduces the subsequent secretion of insulin from the pancreas.
In an experimental investigation, metformin significantly reduced circulating concentrations of glucose and insulin following the administration of a bolus of dextrose by nasogastric tube [24]. There is some unpublished evidence that metformin may reduce insulin concentrations in ponies with EMS but it does not appear to reduce adiposity (Kelly Chameroy, unpublished data). Whether metformin makes a clinically relevant difference to insulin concentration or laminitis risk in the field has not been adequately determined.
Levothyroxine is probably a better alternative for horses with hyperinsulinemia that need to lose weight and for those that do not need to lose weight but have high insulin concentrations, ertugliflozin is likely to be more effective. If metformin is being used, then it is generally administered at 15-30 mg/kg, 2 to 4 times per day. It is likely to be more effective if it can be administered little and often.
Updates on the diagnosis of PPID
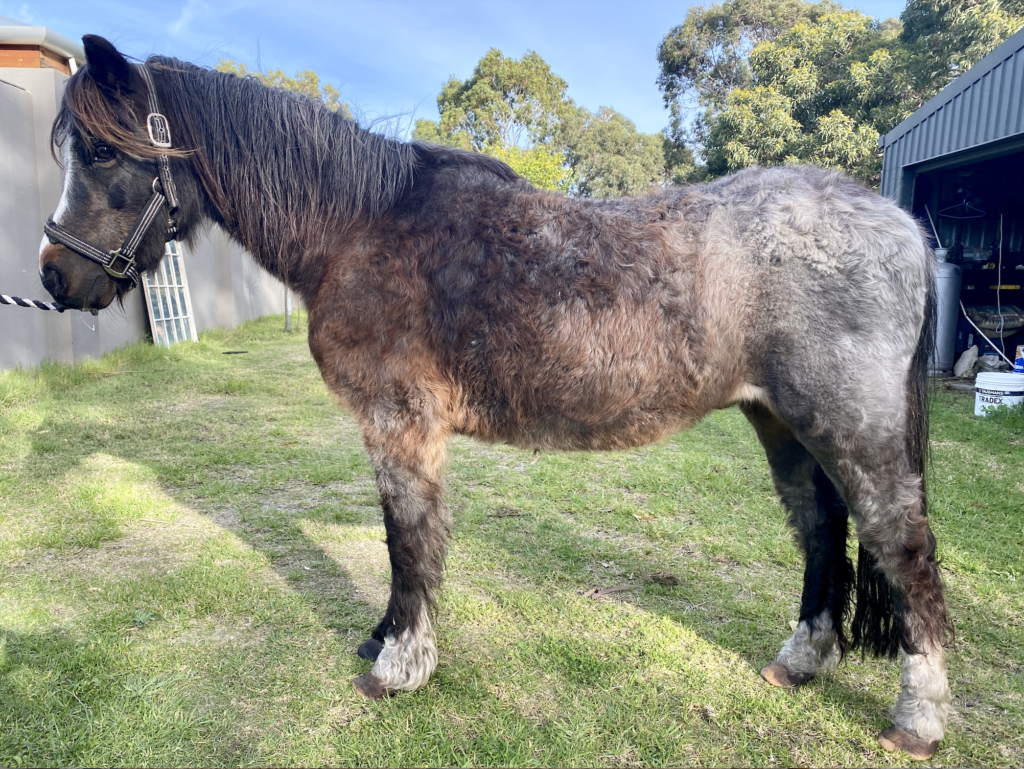
PPID is often stated to be the most common endocrinopathy in aged horses and ponies [25] (Figure 5).
One study in Queensland estimated that one-third of horses were over the age of 16yrs suggesting that PPID may be encountered in practice more frequently given the prevalence of older animals [26]. PPID is a neurodegenerative condition which results in loss of dopaminergic inhibition of the pars intermedia and leads to an overproduction of peptides including adrenocorticotrophic hormone (ACTH) [7]. A high adrenocorticotropic hormone (ACTH) concentration in association with clinical signs provides a diagnosis of PPID.
However, a recent University of Queensland study [27] found that the accuracy of the test was lower in late summer and early autumn which is in contrast to what has been reported previously [28]. The use of ACTH concentration in the absence of clinical signs is not recommended and if clinical signs are not evident then ACTH concentration should be assessed following the administration of thyrotropin releasing hormone (TRH) to increase the accuracy of diagnosis. ACTH concentration can increase in response to many physiological as well as pathological processes and slight increases above normal reference intervals should be interpreted with scepticism and prompt further confirmatory testing. Recent publications have reviewed the diagnosis of PPID [10,28,29].
Updates on the treatment of PPID
Pergolide
Pergolide has been used widely for the treatment of PPID for over 20 years. Most horses exhibit a good improvement in clinical signs and a reduction in ACTH concentration even though in the majority ACTH does not return to normal. Good preventive medicine, farriery, nutrition, dentistry and parasite control are essential in the management of PPID and are as important as pharmaceuticals. Anecdotally pergolide seems to reduce the incidence of laminitis although recent evidence indicates that dopamine agonists do not improve insulin dynamics [30] and there is no evidence that pergolide reduces the risk of laminitis in horses with PPID [31]. PPID and obesity are common co-morbidities and how (or indeed whether) PPID contributes to insulin dysregulation and laminitis risk is not known. A recent study indicated that insulin dysregulation is refractory to treatment in horses with PPID even when they are treated with pergolide (McGowan and Hertzel unpublished data). If there is a concern over laminitis then it is essential that horses with PPID have insulin concentrations assessed and that treatment of concurrent EMS is considered.
In Australia, pergolide is registered in both a tablet (Prascend; Boehringer Ingelheim) and an oral suspension (Pergolide; Ranvet). Some horses are refractory to oral administration and once daily dosing may be challenging for owners. A survey that compared the amount of pergolide used with the amount that should have been dispensed demonstrated that compliance was very poor with only 48% of horses receiving > 90% of the amount they should have taken [32]. This may have implications for the control of clinical signs even though it did not appear to be associated with any difference in ACTH concentration. [31]
Cabergoline
Cabergoline is a dopamine agonist that has the same mechanism of action as pergolide but is used as a long-acting intramuscular injection. It is being used as an alternative to pergolide in the United States as a fortnightly injection and has the potential to facilitate treatment in PPID cases that are difficult to treat daily with an oral medication. Clinical data on the efficacy of cabergoline in the treatment of PPID is lacking but anecdotal reports are promising and there is evidence that it is effective in supressing the output of the pars intermedia (Luoda pharma, unpublished data).
Conclusions
EMS and PPID are two of the most common conditions in equine practice and can have a devastating effect on equine welfare. Interest in both conditions has increased markedly and that interest has been associated with improvements in diagnostic testing and the development of novel treatments that have the potential to improve the control of both conditions. Diet and exercise will always be the mainstay of effective management of EMS and prevention of laminitis; however, ertugliflozin and other insulin sensitising drugs have the potential to reduce insulin concentrations and the incidence of laminitis. They may also promote recovery from laminitis. Levothyroxine offers the additional benefit of promoting weight loss as well as reducing insulin concentrations. In the management of PPID, pergolide remains the mainstay of treatment. Cabergoline offers an interesting and more convenient alternative but clinical data from horses with PPID is limited currently.
Tania Sundra BSc.(Hons) BVMS MANZCVS (Equine Medicine)
Avon Ridge Equine Veterinary Services, Brigadoon, WA, Australia
Tania Sundra graduated from Murdoch University in 2009 and completed an equine internship at a busy performance horse hospital in the USA. Upon returning home she spent a few years working in racetrack and repro clinics in Western Australia. In 2015, Sundra founded Avon Ridge Equine Veterinary Services – an ambulatory equine practice servicing Perth and surrounding regions. In 2019, she successfully completed examinations to become a member of the Australian & New Zealand College of Veterinary Scientists, Equine Medicine chapter. Sundra has a keen interest in all things related to equine medicine, particularly gastroenterology, endocrinology and emergency and critical care.
David Rendle BVSc MVM CertEM(IntMed) DipECEIM FRCVS
RCVS and European Specialist in Equine Internal Medicine
EMT Consulting, Tiverton, Devon, UK
Following graduation from The University of Bristol in 2001, David Rendle worked in farm animal and equine practice before and completing an internship at Liphook Equine Hospital. After a three-year Horserace Betting Levy Board residency at The University of Glasgow and Liphook Equine Hospital, Rendle spent a number of years at Liphook Equine Hospital with a break to work in the Westcountry and Australia. He moved to Rainbow Equine Hospital in Yorkshire where he became a director prior to the hospital’s acquisition by the VetPartners group. In 2019, Rendle returned to his roots and now lives near family on the edge of Exmoor. He works as an independent equine medicine and therapeutics consultant around tending his sheep and is actively involved in clinical research. He has lectured and published on a broad range of topics but has particular interests in gastrointestinal and endocrine diseases. In 2019, Rendle was made a Fellow of the Royal College of Veterinary Surgeons in recognition of his contribution to clinical practice. He is Junior Vice President of the British Equine Veterinary Association and is chair of their health and medicines committee.
References
1. Potter, S., Bamford, N., Harris, P. and Bailey, S. (2017) Incidence of laminitis and survey of dietary and management practices in pleasure horses and ponies in south‐eastern Australia. Aust Vet J 95, 370–374.
2. Asplin, K.E., Sillence, M.N., Pollitt, C.C. and McGowan, C.M. (2007) Induction of laminitis by prolonged hyperinsulinaemia in clinically normal ponies. Vet J 174, 530 535.
3. Laat, M.A.D., McGowan, C.M., Sillence, M.N. and Pollitt, C.C. (2010) Equine laminitis: Induced by 48 h hyperinsulinaemia in Standardbred horses. Equine Vet J 42, 129 135.
4. Karikoski, N.P., Patterson-Kane, J.C., Singer, E.R., McFarlane, D. and McGowan, C.M. (2015) Lamellar pathology in horses with pituitary pars intermediadysfunction. Equine Vet J 48, 472 478.
5. Karikoski, N.P., McGowan, C.M., Singer, E.R., Asplin, K.E., Tulamo, R.M. and Patterson-Kane, J.C. (2015) Pathology of Natural Cases of Equine Endocrinopathic Laminitis Associated With Hyperinsulinemia. Vet Pathol 52, 945 956.
6. Frank, N. and Tadros, E.M. (2014) Insulin dysregulation. Equine Vet J 46, 103–112.
7. McFarlane, D. (2011) Equine Pituitary Pars Intermedia Dysfunction. Vet Clin North Am Equine Pract 27, 93–113.
8. Durham, A., Frank, N., McGowan, C., Menzies-Gow, N., Roelfsema, E., Vervuert, I., Feige, K. and Fey, K. (2019) ECEIM consensus statement on equine metabolic syndrome. J Vet Intern Med 33, 335. http://murdoch.summon.serialssolutions.com/2.0.0/link/0/
9. Durham, A.E., Frank, N., McGowan, C.M., Menzies-Gow, N.J., Roelfsema, E., Vervuert, I., Feige, K. and Fey, K. (2019) ECEIM consensus statement on equine metabolic syndrome. J Vet Intern Med 47, 347.
10. McFarlane, D. (2019) Diagnostic Testing for Equine Endocrine Diseases Confirmation Versus Confusion. Vet Clin North Am Equine Pract 35, 327–338.
11. Potter, S., Bamford, N., Harris, P. and Bailey, S. (2016) Prevalence of obesity and owners’ perceptions of body condition in pleasure horses and ponies in south‐eastern Australia. Aust Vet J 94, 427–432.
12. Menzies-Gow, N.J., Harris, P.A. and Elliott, J. (2017) Prospective cohort study evaluating risk factors for the development of pasture-associated laminitis in the United Kingdom. Equine Vet J 49, 300 306.
13. Meier, A., Laat, M. de, Reiche, D., Fitzgerald, D. and Sillence, M. (2019) The efficacy and safety of velagliflozin over 16 weeks as a treatment for insulin dysregulation in ponies. Bmc Vet Res 15, 65.
14. Meier, A., Reiche, D., Laat, M. de, Pollitt, C., Walsh, D. and Sillence, M. (2018) The sodium-glucose co-transporter 2 inhibitor velagliflozin reduces hyperinsulinemia and prevents laminitis in insulin-dysregulated ponies. Plos One 13, e0203655.
15. Frank, N. (2018) Safety and efficacy of canagliflozin and octreotide for managing insulin dysregulation in horses. In: American College of Veterinary Internal Medicine Forum.
16. Chameroy, K.A. (n.d.) Diagnosis and Management of Horses with Equine Metabolic Syndrome.pdf.
17. Frank, N., Elliott, S.B. and Boston, R.C. (2008) Effects of long-term oral administration of levothyroxine sodium on glucose dynamics in healthy adult horses. American journal of veterinary research 69, 76 81.
18. Frank, N., Elliott, S.B. and Boston, R.C. (2008) Effects of long-term oral administration of levothyroxine sodium on glucose dynamics in healthy adult horses. Am J Vet Res 69, 76–81.
19. Sommardahl, C.S., Frank, N., Elliott, S.B., Webb, L.L., Refsal, K.R., Denhart, J.W. and Thompson, D.L. (2005) Effects of oral administration of levothyroxine sodium on serum concentrations of thyroid gland hormones and responses to injections of thyrotropin-releasing hormone in healthy adult mares. 66, 1025 1031.
20. Tóth, F., Frank, N., Geor, R.J. and Boston, R.C. (2010) Effects of pretreatment with dexamethasone or levothyroxine sodium on endotoxin-induced alterations in glucose and insulin dynamics in horses. Am J Vet Res 71, 60 68.
21. Frank, N., Buchanan, B.R. and Elliott, S.B. (2008) Effects of long-term oral administration of levothyroxine sodium on serum thyroid hormone concentrations, clinicopathologic variables, and echocardiographic measurements in healthy adult horses. Am J Vet Res 69, 68 75.
22. François, R.B., Lauren, E.F. and Janice, E.,K., (2019) Effects of high doses of levothyroxine sodium on serum concentrations of triiodothyronine and thyroxine in horses. Am J Vet Res 80, 565.
23. Durham, A.E. (2017) Therapeutics for Equine Endocrine Disorders. Vet Clin North Am Equine Pract 33, 127 139.
24. Rendle, D.I., Rutledge, F., Hughes, K.J., Heller, J. and Durham, A.E. (2013) Effects of metformin hydrochloride on blood glucose and insulin responses to oral dextrose in horses. Equine Vet J 45, 751 754.
25. Durham, A.E. (2016) Endocrine Disease in Aged Horses. Vet Clin North Am Equine Pract 32, 301–315.
26. McGowan, T., Pinchbeck, G., Phillips, C., Perkins, N., Hodgson, D. and McGowan, C. (2010) A survey of aged horses in Queensland, Australia. Part 1: management and preventive health care. Aust Vet J 88, 420–427.
27. Remona, H.J., Allison, V.J., Karen, L.D., Elizabeth, E., M., Carlos and François‐René, B. (2021) Clinical implications of using adrenocorticotropic hormone diagnostic cutoffs or reference intervals to diagnose pituitary pars intermedia dysfunction in mature horses. J Vet Intern Med 35, 560.
28. Secombe, C., Bailey, S., Laat, M. de, Hughes, K., Stewart, A., Sonis, J. and Tan, R. (2018) Equine pituitary pars intermedia dysfunction: current understanding and recommendations from the Australian and New Zealand Equine Endocrine Group. Aust Vet J 96, 233–242.
29. Tatum, R.C., McGowan, C.M. and Ireland, J.L. (2021) Evaluation of the sensitivity and specificity of basal plasma adrenocorticotrophic hormone concentration for diagnosing pituitary pars intermedia dysfunction in horses: A systematic review. Vet J 105695.
30. Valencia, N.A., Jr, D.L.T. and Oberhaus, E.L. (2017) Long-term and Short-term Dopaminergic (Cabergoline) and Antidopaminergic (Sulpiride) Effects on Insulin Response to Glucose, Glucose Response to Insulin, or Both in Horses. J Equine Vet Sci 59, 95 103.
31. Knowles, E.J. (2018) Does pergolide therapy prevent laminitis in horses diagnosed with pituitary pars intermedia dysfunction? Equine Vet Educ 16, 742 3.
32. Hague, N., Durham, A.E. and Menzies-Gow, N.J. (2020) The effect of pergolide dosing compliance on the laboratory control of piyuitary pars intermedia dysfunction. In: 4th Global Equine Endocrine Symposium , Gut Isling .